C 01 02 cau hinh electron cua nguyen tu va ion
Bạn đang xem bản rút gọn của tài liệu. Xem và tải ngay bản đầy đủ của tài liệu tại đây (1.06 MB, 10 trang )
1
electron
electron
electron
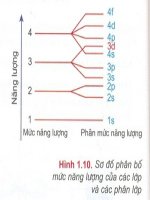
n có n
s
1
2
p
3
6
d
5
10
f
7
14
Obitan (AO)
21
Nguyên lí Pau–li:
VD:
Sai
–
–
–
VD: C (Z = 6)
1s2
2s2
2p2
N (Z = 7):
1s2
2s2
2p3
1
Chú ý
1
(A
(C
(B
(D
2
(A
không
(B
(C
(D
3
(A) 1s 2s 2p 3s 3p 3d 4s …
(C) 1s 2s 2p 3s 3p 4s 3d …
Chú ý
là 2n2
(B) 1s 2s 2p 3s 4s 3p 3d …
(D) 1s 2s 3s 4s 2p 3p 3d …
4
(A) 2.
(B) 6.
(C) 10.
(D) 14.
5
(A) s.
(B) p.
(C) d.
(D) f.
6
(A) 9.
(B) 18.
(C) 6.
(D) 3.
7
(A) 2p.
(B) 3f.
(C) 4d.
không
(D) 5s.
8
(A) 2d.
(B) 3d.
(C) 1p.
(D) 2f.
2
1s 2s 2p 3s 3p 3d 4s 4p …
o
electron và
i n tr c 3d
s
4
1s
2s
2p
3s
3p
3d
4s
4p
VD1:
Z = 6:
1s22s22p2.
Z = 20:
1s22s22p63s23p64s2.
Z = 22:
1s22s22p63s23p63d24s2.
VD2:
2Z N
82
Z
26
2Z N
22
N
30
X là Fe.
22s22p63s23p63d64s2.
VD3:
–
–
1s và 2s
1s22s2; 1s22s22p1; 1s22s22p2; 1s22s22p3; 1s22s22p4; 1s22s22p5; 1s22s22p6
3
Cr: 1s22s22p63s23p63d54s1
Cu: 1s22s22p63s23p63d104s1
44s2
94s2
5
(bán bão hòa) và d10 (bão hòa)
.
1s22s22p63s23p63d64s2
VD: Fe (Z = 26):
64s2
.
phi kim.
1, 2, 3 electron
5, 6, 7 electron
4 electron
phi kim.
.
8 electron
9
(A) 1s22s22p63s23p3.
(B) 1s22s22p63s23p1.
(C) 1s22s22p63s13p2.
(D) 1s22s22p63s23p2.
A
(A) 1s22s22p63s23p5.
(B) 1s22s22p63s23p3.
(C) 1s22s22p63s23p4.
(D) 1s22s22p63s23p2.
B
(A) [Ar]3d94s2.
(B) [Ar]3d84s2.
(C) [Ar]3d104s2.
(D) [Ar]3d104s1.
C
(A) [Ar]3d44s2.
(B) [Ar]3d54s2.
(C) [Ar]3d54s1.
(D) [Ar]3d7.
D
(A) [Ar]3d54s2 và [Ar]3d74s2.
(B) [Ar]3d44s2 và [Ar]3d74s2.
(C) [Ar]3d54s1 và [Ar]3d9.
(D) [Ar]3d54s1 và [Ar]3d74s2.
4
E
(A) 1s22s22p1.
(B) 1s22s22p63s2.
(C) 1s22s22p63s1.
(C) 1s22s22p63s2.
F
(A) 1s22s22p63s23p5.
(B) 1s22s22p63s23p1.
(C) 1s22s22p63s23p2.
(D) 1s22s22p63s2.
G
(A) 1.
(B) 2.
(C) 3.
(D) 4.
(B) 2.
(C) 3.
(D) 4.
H
1
là
(A) 1.
I
(A) 7.
2
(B) 8.
(C) 9.
là
(D) 10.
J
(A) 1s22s22p5.
(B) 1s22s22p6.
(C) 1s22s22p63s2.
(D) 1s22s22p63s23p3.
K
(A) 1s22s22p63s2.
(B) 1s22s22p63s1.
(C) 1s22s22p63s23p1.
(D) 1s22s22p5.
L
(A
(C
(B
(D
M
là 4p5
(A) 80.
(B) 81.
(C) 82.
(D) 96.
5
VD4:
+,
Cl–, Ca2+.
Cl (Z = 17), Ca (Z = 20).
– Na (Z = 11): 1s22s22p63s1
Na+: 1s22s22p6
– Cl (Z = 17): 1s22s22p63s23p5
Cl–: 1s22s22p63s23p6
– Ca (Z = 20): 1s22s22p63s23p64s2
Ca2+: 1s22s22p63s23p6
VD5:
Fe (Z = 26).
2+,
– Cu (Z = 29): 1s22s22p63s23p63d104s1
Fe2+
Cu2+: 1s22s22p63s23p63d9
– Fe (Z = 26): 1s22s22p63s23p63d64s2
Fe2+: 1s22s22p63s23p63d6
+
+
(A) 10.
(B) 11.
+:
1s22s22p6
(C) 22.
6
(D) 23.
22s22p63s1
N
2+
và Cr3+
(A) [Ar]3d9 và [Ar]3d14s2.
(B) [Ar]3d74s2 và [Ar]3d3.
(C) [Ar]3d9 và [Ar]3d3.
(D) [Ar]3d74s2 và [Ar]3d14s2.
6
O
Chú ý
O (Z=8), F (Z=9), Ne (Z=10),
Na (Z=11), Mg (Z=12), Al (Z=13),
S (Z=16), Cl (Z = 17), Ar (Z = 18),
K (Z = 19), Ca (Z=20).
22s22p6?
(A) Mg2+, Na+, F–
(B) Ca2+, K+, Cl–.
(C) Ca2+, K+, F–
(D) Mg2+, K+, Cl–.
P
+,
22s22p6
Y–
là:
(A) Na+, Cl–
(B) Li+, F–, Ne.
(C) Na+, F–
(D) K+, Cl–, Ar.
Q Cation R+ và Y–
23p6.
(A) [Ne]3s23p5 và [Ar]4s1.
(B) [Ne]4s1 và [Ne]3s23p5.
(C) [Ar]4s1 và [Ne]3s23p5.
(D) [Ar]3s23p5 và [Ne]4s1.
R Ion R3+
5
(A) [Ar]3d54s24p1.
(B) [Ar]3d9.
(C) [Ar]3d64s2.
(D) [Ar]3d8.
S
3+
(A) [Ar]3d54s1.
(B) [Ar]3d64s2.
(C) [Ar]3d64s1.
(D) [Ar]3d34s2.
7
– 1AO; p – 3AO, d – 5AO, f – 7AO.
VD7:
Cl (Z = 17) và Fe (Z = 26).
– Cl (Z = 17): 1s22s22p63s23p5
1s2 2s2
2p6
3s2
3p5
– Fe (Z = 26): 1s22s22p63s23p64s2
1s2 2s2
2p6
3s2
3p6
3d6
4s2
VD8:
– C (Z = 6): 1s22s22p2
C có 2
1s2 2s2
2p2
– P (Z = 15): 1s22s22p63s23p3
P có 3
1s2 2s2
2p6
3s2
3p3
T
(A)
(B)
(C)
(D)
8
U
(A)
(B)
(C)
(D)
V
(A) 1.
(B) 2.
(C) 3.
(D) 4.
W
và Ca (Z = 20) là
(A) 1 và 2.
(C) 1 và 0.
(B) 2 và 1.
(D) 1 và 1.
X
(A) He và C.
(C) Mg và S.
(B) O và Ca.
(D) O và S.
Y
(A) Fe.
(B) Al.
(C) P.
(D) Cr.
(B) 2.
(C) 3.
(D) 4.
Z
(A) 5.
9
Câu
1
C
Câu
D
D
Câu
P
C
2
B
E
C
Q
C
3
C
F
B
R
C
4
C
G
C
S
B
5
B
H
C
T
A
6
B
I
C
U
D
7
B
J
C
V
A
8
B
K
D
W
C
9
B
L
B
X
D
A
A
M
A
Y
D
B
D
N
C
Z
D
C
B
O
A
10